
Ytterbium Metal is a fascinating element with many surprising aspects. Dr. Emily Carter, a leading expert in rare earth metals, points out, “Ytterbium Metal is a key player in innovation.” This metal, often overshadowed by its more famous neighbors, contains unique properties that merit attention.
One lesser-known fact is its application in laser technology. Ytterbium Metal's ability to function in solid-state lasers opens new avenues for precision applications. Additionally, it plays a crucial role in certain alloys, enhancing strength and durability. Many overlook its potential in electronics and renewable energy sectors.
Despite its value, public awareness remains low. Misconceptions persist about rare earth elements, including Ytterbium Metal. This lack of understanding can hinder progress in harnessing its benefits. Exploring Ytterbium can foster advancements in various industries, but first, we must acknowledge and reflect on its true importance.

Ytterbium, a member of the lanthanide series, was discovered in the 19th century. It was named after the village of Ytterby in Sweden, where many rare earth minerals were found. This metal is not just an element; it reflects a complex journey of scientific exploration. Early chemists worked diligently to isolate it from ores. Their methods were often rudimentary and imprecise.
The quest to understand ytterbium marked a significant milestone. It helped develop modern chemistry techniques. Scientists struggled with purification techniques. Elemental forms were not easy to obtain. Ytterbium’s properties intrigued them, leading to breakthroughs in various fields. Some applications include lasers and electronics.
While its historical significance is clear, gaps remain in our understanding. Researchers still explore its potential uses today. The path of discovery is filled with trial and error. Each failure led to new knowledge. Ytterbium’s journey continues to inspire curiosity in the realm of science. It’s a reminder of the persistent human spirit in unraveling the mysteries of the natural world.
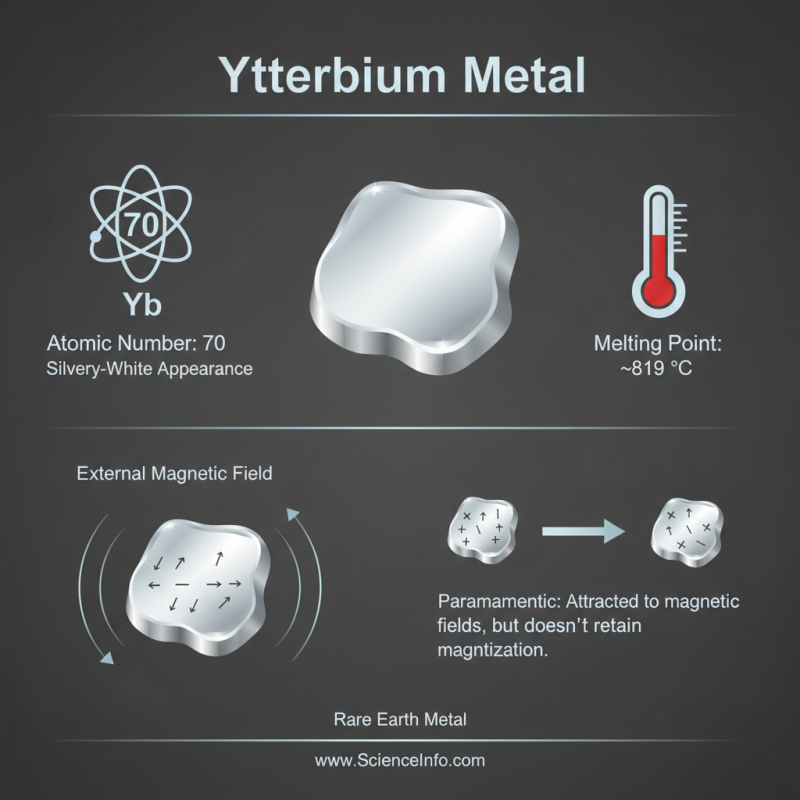
Ytterbium is a rare earth metal, notable for its unique properties. It has an atomic number of 70 and is silvery-white in appearance. Ytterbium exhibits a melting point of about 819 °C, making it relatively stable under high temperatures. This metal is paramagnetic, which means it can be magnetized in the presence of an external magnetic field but does not retain magnetization once the field is removed.
One interesting property of ytterbium is its ability to alter optical properties. It can enhance the performance of certain laser systems. According to a report by the International Journal of Optics, ytterbium-doped lasers are highly efficient and offer a wide wavelength range. These lasers are useful in various applications, including material processing and medical technologies.
Despite its advantages, there are challenges in using ytterbium. The extraction and purification processes can be costly and environmentally taxing. Reports indicate that the global supply of ytterbium may be limited by geological factors and mining practices. This raises questions about the sustainability of sourcing this metal for future technological advancements.
Ytterbium is a rare-earth metal with fascinating uses in technology and industry. It plays a significant role in various fields. One of its notable applications is in fiber-optic technology. Ytterbium-doped fibers improve signal strength in telecommunications. This results in faster and more reliable internet connections. It's essential in high-performance lasers too.
In the healthcare sector, ytterbium is used in medical imaging. Its properties allow for clearer images during scans. This aids in accurate diagnosis and better treatment plans. Ytterbium also presents a useful option for durable materials. It enhances the mechanical properties of alloys, making them stronger and more resistant to wear.
Tips for those interested in ytterbium applications: Research current trends in fiber optics. Understanding its use in communications can open new opportunities. Additionally, explore how it contributes to advancements in medical technology. As industries evolve, the demand for such materials may grow. Stay informed about these developments.
Ytterbium has several isotopes, each with unique properties. The most stable isotope is Ytterbium-174, making up nearly 32.5% of natural ytterbium. Its abundance plays a crucial role in various research fields. Ytterbium-170, on the other hand, is used in medical applications, particularly in radiation therapy. This isotope has a half-life of just over 7 years. Such isotopes exhibit different responses under radiation, making careful choice essential in their applications.
Ytterbium isotopes are prevalent in nuclear reactions. Isotopes with lower atomic mass tend to exhibit better neutron absorption properties. For instance, Yb-173 shows promise for use in nuclear reactors. In terms of practicality, however, the stability and decay rates of these isotopes can complicate their application in technology. Industry reports highlight that managing isotope stability is critical for maintaining safety.
Understanding the differences among these isotopes influences their use in cutting-edge technology. Current research reveals the need for consistent data on their behavior. While Yb-171 may show benefits, its instability limits commercial production. This inconsistency prompts ongoing studies, reflecting the complexity of isotope applications. Insights into these differences can help steer future developments, yet this field still has roadblocks to overcome.
| Fact Number | Fact Description |
|---|---|
| 1 | Ytterbium is a member of the lanthanide series and has the atomic number 70. |
| 2 | It was discovered in 1878 by the Swedish chemist Jean Charles Galissard de Marignac. |
| 3 | Ytterbium is primarily used in the manufacturing of certain laser materials. |
| 4 | It has several isotopes, the most stable being Yb-174 with a half-life of 6.5 hours. |
| 5 | Ytterbium is used in stainless steel and in alloys to enhance their mechanical strength. |
| 6 | It is non-toxic, making it safer compared to some other rare earth metals. |
| 7 | Ytterbium has a relatively low melting point of 824 °C (1515 °F). |
| 8 | It plays a role in various applications, including nuclear medicine for radiological imaging. |
| 9 | Ytterbium can absorb infrared light, making it useful in fiber optic applications. |
| 10 | It is often used as a doping agent in solid-state lasers. |

Handling ytterbium metal requires careful attention to safety and environmental factors. This rare earth element can pose risks if not managed properly. It’s essential to wear appropriate personal protective equipment. Gloves and safety goggles are a must. Ytterbium can cause skin irritation, so direct contact should be avoided.
In a laboratory or industrial setting, proper ventilation is crucial. This minimizes inhalation risks. Ytterbium dust can be harmful if airborne. Spills and leaks should be addressed immediately. It’s wise to have a spill kit ready on-site. Disposal of ytterbium must follow local regulations. Throwing it in regular waste is not safe or responsible.
It is also important to educate all personnel handling ytterbium. Regular training can prevent accidents. Awareness of potential hazards around the metal can lead to better practices. Don’t overlook the importance of clear labeling on containers. Inadequate labeling could lead to confusion and mishandling of materials. Thoughtful approaches can enhance safety and minimize environmental impact.






